by Russell D. White, MD
Type 2 Diabetes Is a Progressive Disease
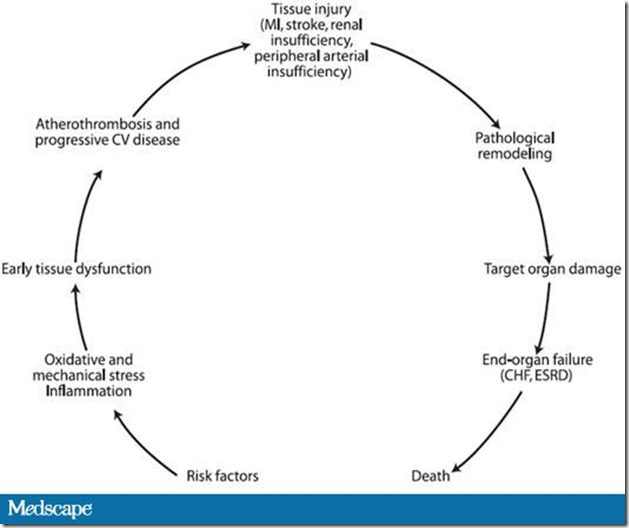
Because of the progressive nature of type 2 diabetes, the majority of patients with longstanding disease are likely to be candidates for insulin therapy.[1]
- This is due to the fact that a large fraction of β-cell function has been lost by the time the disease is diagnosed and this decline continues over time.[2]
- During the early stages of type 2 diabetes, lifestyle interventions may be successful for improving glycemic control for some patients,[3] but as β-cell function continues to decline, pharmacologic treatment is likely to be necessary:
- Oral therapy may progress from treatment with a single agent to combination therapy with 2 or 3 drugs.[4]
- When oral agents are not effective in reaching A1C targets, addition of insulin therapy is required.[5]
- There is an emerging awareness that using insulin earlier in the course of the disease is physiologically sound and should be an integral part of adequate diabetes management.[6]
Progressive Disease Requires Progressive Treatment
Insulin treatment for patients with diabetes should be aimed at treating to target, using information gained from careful blood glucose (BG) monitoring.[7]
- As the disease progresses, so should therapy.
- Treatment should be aimed at a specific A1C goal and adjusted as needed to reach it.
- This approach is in contrast to a slower stepwise approach likely to result in repeated failure and loss of glycemic control.
Basal Insulin
There are multiple approaches to the initiation of insulin therapy in a patient with type 2 diabetes who can no longer maintain glycemic control on oral agents.
- Addition of basal insulin therapy to oral drugs:
- This approach is consistent with results from clinical trials that have shown that adding basal insulin to ongoing oral therapy can improve glycemic control.[8]
- Addition of basal insulin is thought to improve control over BG by suppression of overnight hepatic glucose production, both through direct effects on the liver and indirect effects due to inhibition of free fatty acid release by adipose tissue.[9] Decreasing elevated fasting plasma glucose may decrease the risk for long-term complications of diabetes.[10]
- Main options for basal insulin therapy[11]:
- Intermediate-acting neutral protamine Hagedorn (NPH) insulin
- Long-acting insulin analog (insulin glargine or insulin detemir)
- The pharmacokinetic/pharmacodynamic profiles for NPH insulin and long-acting insulin analogs are substantially different. Those for the long-acting insulin analogs are relatively flat, whereas those for NPH insulin have a distinct peak at approximately 4 hours. This difference results in different dosing requirements for these insulins[9]:
- NPH insulin must be given in the evening or at bedtime and often requires twice daily administration, with a second dose in the morning.
- Long-acting insulin analogs can be administered at bedtime, before dinner, or in the morning, due to their longer durations of action and lower peak effects.
- There are other important differences between NPH insulin and long-acting insulin analogs:
- The pharmacokinetic effects of NPH insulin are more variable than those of insulin glargine or insulin detemir. This variability is a result of the manner in which NPH insulin is structured[12]:
- Protamine is added to regular insulin to extend its duration of action. Addition of protamine increases the tendency of insulin molecules to remain in a hexameric structure at the injection site, resulting in a longer duration of action and longer time to peak. However, the resulting poor solubility of this insulin preparation increases the variability in its pharmacokinetic profile.
- The requirement for resuspension of NPH before injection may also contribute to inter- and intrapatient variability in the action of this insulin because the actual amount of insulin administered may vary from one injection to the next.[12]
- Long-acting insulin analogs have much less variability than NPH. The coefficient of variation for a glucose infusion in patients given 0.4 U/kg NPH insulin is 68%, that for insulin glargine is 48%, and that for insulin detemir is 27%.[13]
- This may contribute to the lower risk for hypoglycemia with these agents versus NPH insulin.[7]
- Clinical studies have demonstrated the effectiveness of adding a long-acting basal insulin analog to oral therapy in patients with type 2 diabetes.[7,14–16]
- Insulin detemir appears to be associated with less weight gain than either insulin glargine or NPH insulin.[17,18]
- The pharmacokinetic effects of NPH insulin are more variable than those of insulin glargine or insulin detemir. This variability is a result of the manner in which NPH insulin is structured[12]:
- How to dose basal insulin:
- The initial dose for insulin detemir is usually 10 U or 0.1-0.2 U/kg.[19]
- In the PREDICTIVE 303 study patients self-adjusted their insulin detemir dose every 3 days based on the average of three self-monitored BG (SMBG) values. Insulin detemir doses were adjusted as follows[20]:
- If the mean fasting plasma glucose (FPG) is <80 mg/dL, the dose is reduced by 3 U.
- If the mean FPG is 80 to 110 mg/dL, there is no change in dosing.
- If the mean FPG is >110 mg/dL, the dose is increased by 3 U.
- One recent study has demonstrated that patients can safely self-titrate insulin detemir to an FPG target of 70-90 mg/dL with low rates of hypoglycemia.[21]
- The typical initial dose for insulin glargine is 10 U.[22] In one study, doses for insulin glargine were adjusted as follows[23]:
- No change is required if FPG remains between 70 and 94 mg/dL.
- If FPG is <70 mg/dL for 3 days, the dose should be decreased by up to 10% of the total dose.
- If FPG is 95 to 119, 120 to 139, 140 to 180, or >180 mg/dL for 3 days, the dose should be increased by 2, 4, 6, or 8 U, respectively.
Adding Prandial Insulin
While addition of basal therapy is highly effective in many patients, the progressive nature of diabetes may require further intensification of treatment. Rapid-acting insulin analogs are a suitable first choice for intensification of therapy:
- Rapid-acting insulin analogs have lower variability in absorption and more consistent pharmacodynamic profiles than regular human insulin.[24,25]
- Rapid-acting insulin analogs provide higher 1- and 2-hour insulin values, reduced risk for late postprandial hypoglycemia due to a shorter duration of action than regular human insulin, and may provide quality of life benefits due to greater flexibility in timing and meal-time dosing.[26]
- These insulin analogs provide a more physiologic action which coincides with meal patterns.
Adding prandial insulin to basal insulin may be the best way to restore postprandial and overall glycemic control when the combination of basal insulin and oral therapies is no longer effective. Prandial insulin may be added to basal insulin in a step-wise manner[9]:
- Adding prandial insulin to decrease postprandial glucose (PPG) may be most beneficial in patients with FPG that is at or near goal, but who still have modestly elevated A1C. Results from Monnier and colleagues have shown that PPG makes the greatest contribution to A1C at values below approximately 8.5%.[27] Addition of prandial insulin should also be considered when the basal insulin dose is very high (>0.6 U/kg).
- A single mealtime prandial injection of a rapid-acting insulin analog that controls the highest postprandial glucose (PPG) may be sufficient to restore glycemic control[28]:
- The basal insulin dose should be lowered by the amount of prandial insulin to decrease the risk for nocturnal hypoglycemia.[29]
- The prandial insulin dose can then be titrated in accordance with SMBG measured 2 hours after the start of the meal, before the next meal, or at bedtime if the injection is administered before the evening meal:
- The American Diabetes Association goal for peak PPG is <180 mg/dL.[30]
- The American College of Endocrinology recommends a 2-hour postprandial BG level <140 mg/dL.[31]
- It has also been shown that prandial insulin doses can be effectively adjusted based on premeal BG patterns from the previous week.[23]
- The prandial insulin dose can be adjusted independently to limit postprandial hyperglycemia without affecting basal insulin action.[9,27]
Basal-Bolus Therapy
Basal-bolus therapy may be an appropriate treatment progression for patients who have had prandial insulin added to the treatment regimen[32]:
- In these regimens, basal and bolus insulin requirements are each approximately 50% of the total daily insulin needed.
- Results from the PREFER study indicated that one-third of the total prandial insulin dose can be delivered with each meal with the majority of patients achieving A1C ≤7%.[33]
- It has also been suggested that the total dose of rapid-acting insulin analog may be divided with 38% delivered at breakfast, 28% at lunch, and 33% at dinner (White RD, et al., unpublished data). The highest prandial dose is delivered at breakfast for two reasons:
- High carbohydrate content for this meal
- The “dawn phenomenon,” a morning surge in plasma glucose that occurs secondary to a physiologic morning rise in cortisol and growth hormone levels.[34]
There are several considerations for titration of prandial insulin:
- Rapid-acting insulin analogs have accelerated pharmacokinetics compared with long-acting insulins, and they require different adjustments that can be made at shorter intervals.
- SMBG should be carried out at least three times per day for patients using multiple insulin injections.[30]
- Exercise improves insulin sensitivity, and it is often necessary to reduce the prandial insulin before exercise occurring within 3 hours in patients who engage in moderate or strenuous exercise.[35] Carbohydrate counting is an alternative to dosage algorithms for determining prandial insulin dose, and it provides for more flexibility in meal planning,[23] but this is not absolutely necessary.
- Concern about hypoglycemia is a significant psychological barrier to intensive insulin therapy[36]:
- This risk is reduced by the use of long- and rapid-acting insulin analogs versus human insulins.
- In one study, switching patients from the combination of regular human insulin and NPH insulin to insulin lispro and insulin glargine resulted in a 44% reduction in the occurrence of hypoglycemia.[37]
- A second study indicated a 43% reduction in the occurrence of nighttime hypoglycemia with the insulins lispro and glargine versus regular human and NPH insulin.[38]
- The combination of insulins detemir and aspart has also been shown to be associated with 38% lower risk for nighttime hypoglycemia than NPH plus regular human insulin in patients receiving basal-bolus treatment.[39]
Simplifying Basal-Bolus Therapy
Advances in therapy may help to simplify basal-bolus therapy:
- Rapid-acting insulin analogues that can be administered shortly before or even after meals[40] have the potential to improve adherence.
- Adherence to treatment in difficult-to-manage patients may also be improved with intensive home- and community-based family therapy.[41]
- Patient referral to specialists for intensive diabetes care may be necessary to reach treatment targets in some patients.[42]
- Use of an insulin pen may improve adherence to treatment and thus glycemic control. It has been shown that patients with type 2 diabetes prefer insulin pen devices over syringe and vial; and these devices improve adherence to therapy and decrease the risk for hypoglycemia.[43]
- Additional steps that may increase the probability of reaching treatment goals include[44]:
- Monitoring A1C every 3 months in addition to SMBG
- Aggressively managing hyperglycemia, dyslipidemia, and hypertension with the same intensity to obtain the best patient outcome
- Implementing a multi- and interdisciplinary team approach to diabetes management both to encourage patient education and self-care and to share responsibility for patients achieving glucose goals
References
- Wright A, Burden AC, Paisey RB, Cull CA, Holman RR, for the U.K. Prospective Diabetes Study Group. Sulfonylurea inadequacy. Efficacy of addition of insulin over 6 years in patients with type 2 diabetes in the U.K. Prospective Diabetes Study (UKPDS 57). Diabetes Care. 2002;25:330-336.
- DeFronzo RA. From the triumvirate to the ominous octet – a new paradigm for the treatment of T2DM. The Banting Lecture. Programs and abstracts of the American Diabetes Association 68th Scientific Sessions; June 6-10, 2008; San Francisco, California.
- Petersen KF, Dufour S, Befroy D, Lehrke M, Hendler RE, Shulman GI. Reversal of nonalcoholic hepatic steatosis, hepatic insulin resistance, and hyperglycemia by moderate weight reduction in patients with type 2 diabetes. Diabetes. 2005;54:603-608.
- Bell DS. Type 2 diabetes mellitus: what is the optimal treatment regimen? Am J Med. 2004;116(suppl 5A):23S-29S.
- Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy. A consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2009;32:193-203.
- Eldor R, Stern E, Milicevic Z, Raz I. Early use of insulin in type 2 diabetes. Diabetes Res Clin Pract. 2005;68(suppl 1):S30-S35.
- Riddle MC, Rosenstock J, Gerich J; on behalf of the Insulin Glargine 4002 Study Investigators. The treat-to-target trial. Randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients. Diabetes Care. 2003;26:3080-3086.
- Davies M, Evans R, Storms F, Gomis R, Khunti K; on behalf of the AT.LANTUS Study Group. Initiation of insulin glargine in suboptimally controlled patients with type 2 diabetes: sub-analysis of the AT.LANTUS trial comparing treatment outcomes in subjects from primary and secondary care in the UK. Diabetes Obes Metab. 2007;9:706-713.
- Raccah D, Bretzel RG, Owens D, Riddle M. When basal insulin therapy in type 2 diabetes mellitus is not enough–what next? Diabetes Metab Res Rev. 2007;23:257-264.
- Gimeno-Orna JA, Castro-Alonso FJ, Boned-Juliani B, Lou-Arnal LM. Fasting plasma glucose variability as a risk factor of retinopathy in Type 2 diabetic patients. J Diabetes Complications. 2003;17:78-81.
- Scholtz HE, Pretorius SG, Wessels DH, Becker RH. Pharmacokinetic and glucodynamic variability: assessment of insulin glargine, NPH insulin and insulin ultralente in healthy volunteers using a euglycaemic clamp technique. Diabetologia. 2005;48:1988-1995.
- Takiya L, Dougherty T. Pharmacist’s guide to insulin preparations: a comprehensive review. Pharmacy Times CE. 2005. Available at: https://secure.pharmacytimes.com/lessons/200510-03.asp.
- Heise T, Nosek L, Rønn BB, et al. Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes. 2004;53:1614-1620.
- Garber AJ, Clauson P, Pedersen CB, Kølendorf K. Lower risk of hypoglycemia with insulin detemir than with neutral protamine Hagedorn insulin in older persons with type 2 diabetes: a pooled analysis of phase III trials. J Am Geriatr Soc. 2007;55:1735-1740.
- Fritsche A, Schweitzer MA, Häring HU; and the 4001 Study Group. Glimepiride combined with morning insulin glargine, bedtime neutral protamine Hagedorn insulin, or bedtime insulin glargine in patients with type 2 diabetes. A randomized, controlled trial. Ann Intern Med. 2003;138:952-959.
- Hermansen K, Davies M, Derezinski T, Martinez Ravn G, Clauson P, Home P; on behalf of the Levemir Treat-to-Target Study Group. A 26-week, randomized, parallel, treat-to-target trial comparing insulin detemir with NPH insulin as add-on therapy to oral glucose-lowering drugs in insulin-naive people with type 2 diabetes. Diabetes Care. 2006;29:1269-1274.
- Rosenstock J, Davies M, Home PD, Larsen J, Koenen C, Schernthaner G. A randomised, 52-week, treat-to-target trial comparing insulin detemir with insulin glargine when administered as add-on to glucose-lowering drugs in insulin-naive people with type 2 diabetes. Diabetologia. 2008;51:408-416.
- Fakhoury W, Lockhart I, Kotchie RW, Aagren M, LeReun C. Indirect comparison of once daily insulin detemir and glargine in reducing weight gain and hypoglycaemic episodes when administered in addition to conventional oral anti-diabetic therapy in patients with type-2 diabetes. Pharmacology. 2008;82:156-163.
- Levemir. [prescribing information]. 2007. Available at: http://www.levemir-us.com/prescribing_information.pdf. Accessed May 21, 2009.
- Selam JL, Koenen C, Weng W, Meneghini L. Improving glycemic control with insulin detemir using the 303 algorithm in insulin naïve patients with type 2 diabetes: a subgroup analysis of the US PREDICTIVE 303 study. Curr Med Res Opin. 2008;24:11-20.
- Blonde L, Merilainen M, Karwe V, Raskin P; TITRATE Study Group. Patient-directed titration for achieving glycaemic goals using a once-daily basal insulin analogue: an assessment of two different fasting plasma glucose targets - the TITRATE study. Diabetes Obes Metab. 2009;11:623-631.
- Lantus. [prescribing information]. 2007. Available at: http://products.sanofi-aventis.us/lantus/lantus.html. Accessed May 21, 2009.
- Bergenstal RM, Johnson M, Powers MA, et al. Adjust to target in type 2 diabetes: comparison of a simple algorithm with carbohydrate counting for adjustment of mealtime insulin glulisine. Diabetes Care. 2008;31:1305-1310.
- Becker RH, Frick AD. Clinical pharmacokinetics and pharmacodynamics of insulin glulisine. Clin Pharmacokinet. 2008;47:7-20.
- Guerci B, Sauvanet JP. Subcutaneous insulin: pharmacokinetic variability and glycemic variability. Diabetes Metab. 2005;31:4S7-4S24.
- Rossetti P, Porcellati F, Bolli GB, Fanelli CG. Prevention of hypoglycemia while achieving good glycemic control in type 1 diabetes. The role of insulin analogs. Diabetes Care. 2008;31(suppl 2):S113-S120.
- Monnier L, Lapinski H, Colette C. Contributions of fasting and postprandial plasma glucose increments to the overall diurnal hyperglycemia of type 2 diabetic patients Variations with increasing levels of HbA(1c). Diabetes Care. 2003;26:881-885.
- Monnier L, Colette C. Addition of rapid-acting insulin to basal insulin therapy in type 2 diabetes: indications and modalities. Diabetes Metab. 2006;32:7-13.
- Meneghini L. Why and how to use insulin therapy earlier in the management of type 2 diabetes. South Med J. 2007;100:164-174.
- American Diabetes Association. Standards of medical care in diabetes–2009.Diabetes Care. 2009;32(suppl 1):S13-S61.
- American College of Endocrinology. American College of Endocrinology Consensus Statement on Guidelines for Glycemic Control. Endocrinology. 2002;8(suppl 1):6-11.
- Mayfield JA, White RD. Insulin therapy for type 2 diabetes: rescue, augmentation, and replacement of beta-cell function. Am Fam Physician. 2004;70:489-500.
- Liebl A, Prager R, Binz K, Kaiser M, Bergenstal R, Gallwitz B; PREFER Study Group. Comparison of insulin analogue regimens in people with type 2 diabetes mellitus in the PREFER Study: a randomized controlled trial. Diabetes Obes Metab. 2009;11:45-52.
- Carroll MF, Schade DS. The dawn phenomenon revisited: implications for diabetes therapy. Endocr Pract. 2005;11:55-64.
- Banting and Best Diabetes Centre. Approach to the Management of Diabetes Mellitus. 6th ed. Developed by the Diabetes Care and Education Committee, Banting and Best Diabetes Centre. Toronto: Faculty of Medicine, University of Toronto; 2005.
- Hartman I. Insulin analogs: impact on treatment success, satisfaction, quality of life, and adherence. Clin Med Res. 2008;6:54-67.
- Ashwell SG, Amiel SA, Bilous RW, et al. Improved glycaemic control with insulin glargine plus insulin lispro: a multicentre, randomized, cross-over trial in people with Type 1 diabetes. Diabet Med. 2006;23:285-292.
- Murphy NP, Keane SM, Ong KK, et al. Randomized cross-over trial of insulin glargine plus lispro or NPH insulin plus regular human insulin in adolescents with type 1 diabetes on intensive insulin regimens. Diabetes Care. 2003;26:799-804.
- Raslová K, Bogoev M, Raz I, Leth G, Gall MA, Hâncu N. Insulin detemir and insulin aspart: a promising basal-bolus regimen for type 2 diabetes. Diabetes Res Clin Pract. 2004;66:193-201.
- Garg SK, Rosenstock J, Ways K. Optimized basal-bolus insulin regimens in type 1 diabetes: insulin glulisine versus regular human insulin in combination with basal insulin glargine. Endocr Pract. 2005;11:11-17.
- Ellis DA, Frey MA, Naar-King S, Templin T, Cunningham P, Cakan N. Use of multisystemic therapy to improve regimen adherence among adolescents with type 1 diabetes in chronic poor metabolic control. A randomized controlled trial. Diabetes Care. 2005;28:1604-1610.
- Graber AL, Elasy TA, Quinn D, Wolff K, Brown A. Improving glycemic control in adults with diabetes mellitus: shared responsibility in primary care practices. South Med J. 2002;95:684-690.
- Goldstein HH. Pen devices to improve patient adherence with insulin therapy in type 2 diabetes. Postgrad Med. 2008;120:172-179.
- Del Prato S, Felton AM, Munro N, et al; on behalf of the Global Partnership for Effective Diabetes Management. Improving glucose management: ten steps to get more patients with type 2 diabetes to glycaemic goal. Int J Clin Pract. 2005;59:1345-1355.